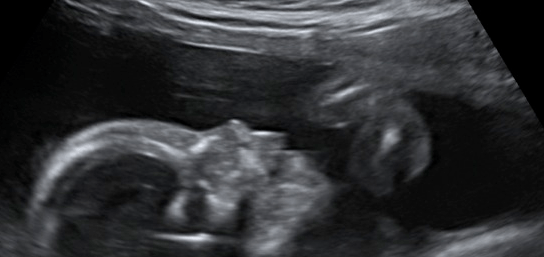
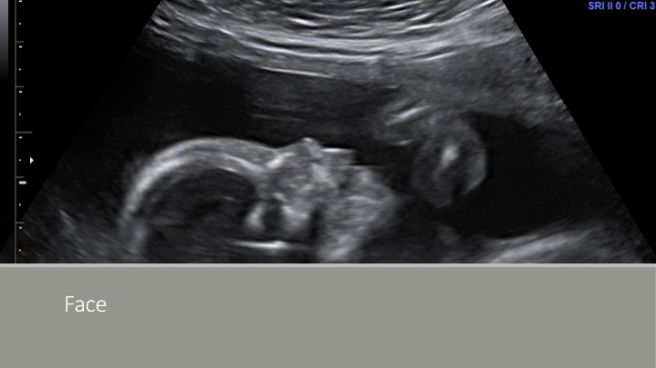
A recent case of severe toxoplasma infection in pregnancy at one of my institutions reinforces the need for preconception and early pregnancy hygiene advice.
What is toxoplasma infection?
Toxoplasmosis is an infection caused by the parasite Toxoplasma gondii. The parasite may be found in raw and cured meat, raw eggs, unpasteurised milk and cat faeces. It can also be found in soil contaminated with cat faeces. This means cat owners need to be careful when they handle kitty litter and should wear gloves when they garden in areas where the soil may be contaminated.
Clinical consequences of toxoplasma infection
Outside of pregnancy, toxoplasmosis results in a mild flu-like illness or a subclinical infection with no symptoms.
However, toxoplasma infection acquired in the period from three months prior to conception and during pregnancy, is more significant. The parasite can cross the placenta and infect the developing baby who has an immature immune system. The consequences can be serious and include miscarriage, brain damage, blindness and death.
Why the timing of the infection is important
The timing of the infection in pregnancy influences the risk of the baby acquiring the infection. It also impacts on the severity of the sequelae of the infection in the baby.
In early pregnancy, less than 5% of babies are infected. This is because the placenta is too immature for transplacental infection.
However, in late pregnancy, 65% of babies can acquire the infection, as the placenta is mature and rates of transplacental infection are higher.
Even though infection caught late in pregnancy is more likely to result in fetal infection, the consequences of the infection are usually less severe.
In contrast, infection caught by the mother early in her pregnancy results in a lower fetal infection rate but has far more significant consequences for her baby.
Most of the advice on toxoplasma in pregnancy comes from a few landmark research papers. It is worth looking at the findings from these papers to help guide pregnancy advice and management.
Landmark research papers
1. Pregnancy outcomes
The first landmark paper on toxoplasma in pregnancy was published in the New England Journal of Medicine in 1974 (1).
The researchers reported on the pregnancy outcomes of two groups of women. One group had evidence of toxoplasmosis prior to pregnancy and the other group acquired toxoplasmosis during pregnancy.
The key findings were:
- Mothers who acquired toxoplasmosis prior to pregnancy did not infect their baby. No babies in this group had toxoplasmosis.
However;
- In the 77 women who acquired toxoplasma infection during pregnancy:
- 11 women miscarried;
- 7 women suffered fetal death in utero and delivered a stillborn baby;
- 2 newborns died soon after delivery;
- 7 babies had severe effects of congenital toxoplasmosis and had cerebral and ocular complications;
- 11 babies had only mild illness requiring treatment:
- 39 babies had subclinical illness with no long term problems.
- Severe outcomes were only detected when the maternal toxoplasma infection occured during the first two trimesters (up to 27 weeks pregnancy).
- Acquiring the toxoplasma infection in the third trimester resulted in subclinical infection or in no fetal infection.
2. Risk factors
The largest published research paper on risk factors comes from France (2), which has one of the highest rates of toxoplasma infection in pregnancy in the world. The authors reported that there were approximately 4900 cases of primary Toxoplasma infection in pregnant women in France each year.
Since 1992, France has had a policy that all pregnant women at risk of Toxoplasma infection undergo monthly serological testing. This research paper reported on 80 pregnant women who seroconverted to Toxoplasma in pregnancy and compared their risk factors against 80 pregnant women who had repeatedly negative tests.
After performing a multivariate statistical analysis to ensure differences between the two groups of women were accounted for, they found the risk factors for Toxoplasma infection were:
- Poor hand hygiene increased risk by almost 10 fold (OR=9.9; 95%CI: 0.8-125);
- Consumption of undercooked beef increased risk by almost 6 fold (OR=5.5; 95%CI: 1.1-27);
- Having a pet cat increased risk by almost 5 fold (OR=4.5; 95%CI: 1.0-19.9);
- Frequent consumption of raw vegetables outside the home increased risk by 3 fold (OR=3.1; 95%CI: 1.2-7.7);
- Consumption of undercooked lamb increased risk by 3 fold (OR=3.1; 95%CI: 0.85-14).
The researchers noted that when women were provided with documentary advice on how to prevent toxoplasma infection, they had a lower risk of infection. They concluded that all pregnant women should be given information on their eating habits, hand hygiene and cats in order to reduce the risk of acquiring toxoplasma infection in pregnancy.
3. Treatment of toxoplasma infection in pregnancy
The third landmark paper addressed the treatment of toxoplasma infection in pregnancy (3).
This treatment study was conducted across 5 major centres that specialised in the management of toxoplasma infection in pregnancy. The aim of the study was to determine if prenatal antibiotic therapy could reduce the rate of mother to baby transmission of Toxoplasma gondii and reduce the risk of severe consequences in infected babies as measured when they were one year old.
The study followed 144 women and considered key factors such as the gestational age at which the infection was acquired, the administration of antibiotic therapy, duration of antibiotic therapy, and time lapse between infection and the start of antibiotic therapy.
The authors reported that 64 of the 144 infected women (44%) gave birth to a congenitally infected infant.
After performing a multivariate analysis they concluded that the rate of transmission from the mother to the baby was not affected by the administration of antibiotics. The only factor that affected the rate of transmission was the gestational age at which the mother developed the infection in pregnancy (P < .0001).
However, antibiotic administration significantly reduced the risk of serious sequelae in the babies (P = 0.026, odds ratio 0.30, 95% confidence interval 0.104-0.863).
Of particular note, severe sequelae were significantly reduced when the mother was administered antibiotics (P = .007, odds ratio 0.14, 95% confidence interval 0.036-0.584). The sooner antibiotics were given after the infection, the less frequently sequelae were seen (P = 0.021).
The results were consistent with the earlier New England Journal of Medicine paper which also reported that treatment of infected mothers with the antibiotic Spiramycin reduced the impact of fetal infection (1).
What is the current pregnancy advice for preventing toxoplasmosis in pregnancy?
In Australia, the risk of toxoplasma infection is low. Therefore routine screening for the infection is not performed.
The main method to prevent infection is through education on strategies to reduce the risk of becoming infected.
The Australian Government’s Pregnancy, Birth and Baby website outlines strategies to reduce the risk of infection (4). These are:
- Wear gloves when gardening, particularly when handling soil;
- Wash your hands thoroughly after handling soil, using soap and hot water;
- Do not eat raw or undercooked meat;
- Cook all red meat until no trace of pinkness remains and the juices run clear;
- Do not eat cured meats (e.g. Parma ham, salami);
- Do not taste meat before it is fully cooked;
- Wash your hands thoroughly after handling raw meat;
- Wash all kitchenware thoroughly after preparing raw meat;
- Always wash fruit and vegetables before cooking and eating;
- Avoid drinking unpasteurised goat’s milk or eating products that are made from it;
- Do not handle or adopt stray cats;
- Keep animals, especially cats, away from areas that you prepare/store food;
- Wash your hands and other exposed body parts with soap and running water after touching animals, their enclosures or food containers;
- Avoid cat faeces in cat litter or soil – wear gloves if you are changing a cat’s litter tray and wash your hands thoroughly afterwards. If you are pregnant or immune deficient, ask someone else to change it for you and empty the litter tray daily;
- Feed your cat dried or canned cat food rather than raw meat;
- Cover children’s outdoor sand boxes to prevent cats using them as litter boxes;
- Avoid contact with sheep and newborn lambs during the lambing season.
Management if infection is suspected
If a pregnant woman is concerned that she may have acquired toxoplasmosis in pregnancy, she should immediately see her doctor or midwife to discuss her concerns and be referred for an antibody test.
If the test demonstrates the presence of IgG antibodies, she can usually be reassured that she has already had the infection and is immune.
If the test demonstrates IgM antibodies, or no antibodies but the clinical suspicion of infection is high, the pregnancy should be managed by an obstetrician with expertise in this area.
Many cases are not detected until an ultrasound examination is performed and is noted to be abnormal.
Once maternal infection is diagnosed, or an ultrasound examination is suspicious of features of toxoplasma infection in the fetus, a tertiary level ultrasound, blood tests and amniocentesis to diagnose fetal infection are indicated. Antibiotic therapy needs to be commenced as soon as possible to reduce the impact of the fetal infection.
All cases should be managed in a tertiary obstetric hospital and advice and management will be individualised to each woman.
Babies should be enrolled in a follow up service to ensure any sequelae are diagnosed and managed as soon as possible.
Summary
Toxoplasma infection in pregnancy resulting in severe consequences for the baby is rare. However it can be prevented with good hygiene practices and managed with expert interventions by specialist teams.
The most important message is to be aware of good hand hygiene practices.
References
- Dismounts G, Couvreur J. Congenital Toxoplasmosis — A Prospective Study of 378 Pregnancies.N Engl J Med 1974; 290:1110-1116May 16, 1974. DOI: 10.1056/NEJM197405162902003
- Baril L, Ancelle T, Goulet V, Thulliez P, Tirard-Fleury V, Carme B. Risk Factors for Toxoplasma Infection in Pregnancy: A Case-Control Study in France.
Scandinavian Journal of Infectious Diseases 1999; 31(3): 305-309.
doi.org/10.1080/00365549950163626 -

 Gestational diabetes is a common medical complication of pregnancy (1-5). It is an important condition as failure to diagnose and treat gestational diabetes can lead to poor pregnancy outcomes, and in severe cases, fetal death in utero may occur. However, with accurate diagnosis and management, outcomes are excellent.
Gestational diabetes is a common medical complication of pregnancy (1-5). It is an important condition as failure to diagnose and treat gestational diabetes can lead to poor pregnancy outcomes, and in severe cases, fetal death in utero may occur. However, with accurate diagnosis and management, outcomes are excellent.